The FDA Unleashes Groundbreaking Drug to Combat Life-Threatening Food Allergies!
SUBSCRIBE TO OUR BLOG
Promotions, new products, and recipes.
Food allergies are a serious and potentially life-threatening condition that affects millions of people around the world. The recent tragic incident involving the doctor who passed away after consuming food at Disneyland serves as a stark reminder of the dangers posed by food allergies.
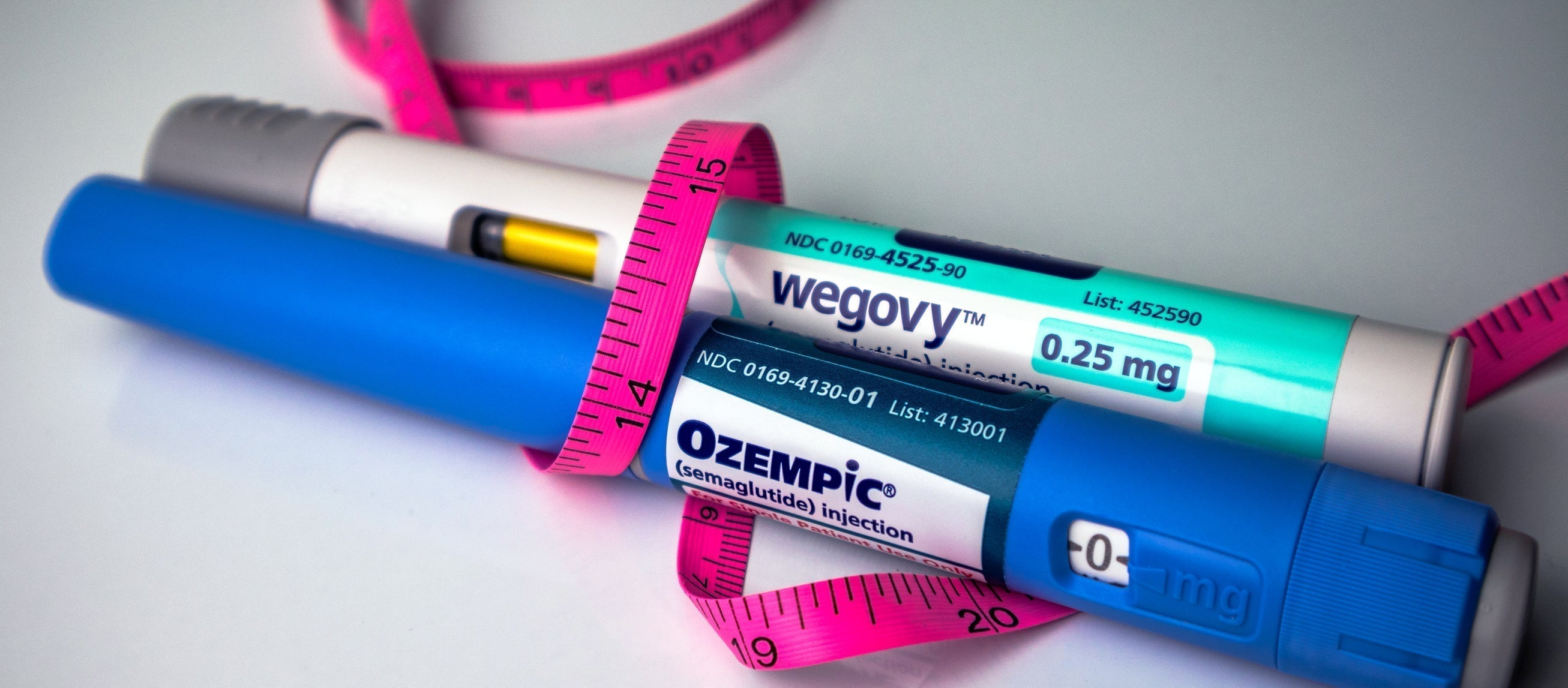
A new study led by scientists at Stanford School of Medicine has found that the drug omalizumab, fda approved medication for severe food allergies, originally approved by the FDA for treating diseases like allergic asthma and chronic hives, can protect people from severe allergic responses to small quantities of allergenic foods. This breakthrough could provide a layer of protection for individuals with food allergies in case of accidental exposure. The study included 177 children who were severely allergic to peanuts and other foods, and after receiving monthly or bimonthly omalizumab injections for four months, two-thirds of the participants could safely consume small amounts of their allergy-triggering foods.
Key Takeaways
- The FDA has approved omalizumab, a groundbreaking drug for xolair use in treating food allergies.
- Omalizumab has demonstrated its safety and effectiveness in treating severe food allergies.
- The drug allows individuals with food allergies to consume small amounts of their allergenic foods without severe allergic reactions.
- This breakthrough offers new hope for those living with life-threatening food allergies.
- Omalizumab could significantly improve the quality of life for individuals with food allergies.
Omalizumab: A Game-Changing Drug for Food Allergy Treatment
Omalizumab, also known by the brand name Xolair, is a FDA-approved drug that has revolutionized the treatment of severe asthma, chronic hives, and nasal polyps. It works by targeting and inactivating the antibodies responsible for triggering allergic reactions, providing relief and improved quality of life for patients. But its impact doesn't stop there. A recent study conducted at Stanford School of Medicine has uncovered the remarkable safety and effectiveness of omalizumab in reducing the risk of allergic reactions to food, paving the way for a groundbreaking approach to food allergy treatment.
With the prevalence of food allergies on the rise, finding safe and effective treatment options has become a pressing concern. Omalizumab has shown great promise in preventing severe allergic symptoms and offering hope to individuals who previously faced the constant fear of accidental food exposure. This drug has the potential to be a game-changer in the field of food allergy treatment, providing a new level of protection and freedom for those with severe allergies.
The Safety and Effectiveness of Omalizumab
The recent study conducted at Stanford School of Medicine revealed the safety and effectiveness of omalizumab in reducing the risk of allergic reactions to food. The study included 177 children severely allergic to peanuts and other foods, and after receiving omalizumab injections for four months, two-thirds of the participants were able to safely consume small amounts of their allergy-triggering foods. These results are statistically significant and highlight the potential of omalizumab to prevent severe allergic symptoms caused by food allergens.
Omalizumab's safety profile has already been well-established through its FDA approval for treating severe asthma, chronic hives, and nasal polyps. As with any medication, there can be potential side effects, and it is important for patients to discuss their medical history and any concerns with their healthcare provider before starting omalizumab treatment. However, the overall safety and effectiveness of omalizumab make it a promising option for individuals with severe food allergies.
Expanding Treatment Options for Food Allergies
The approval of omalizumab for food allergy treatment offers a beacon of hope for individuals with severe allergies. It expands the range of treatment options beyond allergen avoidance and emergency management, providing a proactive approach to preventing severe allergic reactions. Omalizumab's ability to target and inactivate the antibodies responsible for allergic responses makes it a powerful tool in the fight against food allergies.
This drug is particularly significant for individuals who face life-threatening reactions to even trace amounts of allergenic foods. By reducing their risk of severe allergic symptoms, omalizumab empowers these individuals to lead more fulfilling lives without the constant fear of accidental exposures. It opens up new possibilities for socializing, dining out, and participating in everyday activities that were once riddled with anxiety and trepidation.
In the next section, we will explore the prevalence and impact of food allergies in the United States, shedding light on the urgent need for effective treatment options to mitigate the significant burden experienced by allergy sufferers. Stay tuned!
Prevalence and Impact of Food Allergies in the US
Food allergies affect approximately 8% of children and 10% of adults in the United States. These allergies can have a significant impact on individuals' social, psychological, and economic well-being. Accidental exposures to allergens can lead to life-threatening reactions, and individuals with food allergies often face challenges in everyday activities such as attending parties and eating at restaurants.
The FDA's approval of omalizumab for food allergy treatment could provide new options for managing the disease and reducing its health impact. By utilizing new food allergy treatments, such as xolair, individuals may be able to effectively minimize the risks associated with severe allergies, improving their quality of life and allowing them to engage in daily activities without constant fear of life-threatening reactions.
The Science Behind Omalizumab's Effectiveness
Omalizumab, the active ingredient in the drug Xolair, has shown remarkable effectiveness in treating food allergies. Its mechanism of action revolves around targeting immunoglobulin E (IgE), the molecule responsible for triggering allergic reactions in the body.
When administered, omalizumab binds to IgE present in the blood and immune cells, effectively deactivating its ability to elicit an allergic response. This prevents the release of inflammatory chemicals, ultimately reducing the severity of allergic symptoms.
Clinical trials have highlighted the positive outcomes of using omalizumab for the treatment of IgE-mediated food allergies, including severe peanut allergies. These studies have consistently demonstrated a significant reduction in allergic reactions to food allergens in participants who received omalizumab treatment.
Of particular note is its potential as a treatment option for children with allergies, especially those who are at high risk of accidental food exposures. The use of omalizumab in this population has shown promising results in minimizing the risk of severe allergic symptoms and improving their quality of life.
Overall, the science behind omalizumab's effectiveness lies in its ability to target IgE, preventing the cascade of events that lead to allergic reactions. This breakthrough in food allergy treatment offers hope for individuals suffering from severe allergies and paves the way for improved management strategies.
The Role of Omalizumab in Allergen Avoidance
Omalizumab treatment plays a significant role in helping individuals with food allergies avoid allergen exposure. By reducing the risk of severe allergic reactions, omalizumab provides a layer of protection against accidental food exposure, enabling individuals to engage in everyday activities without constant fear of allergen ingestion. This groundbreaking drug, also known as Xolair, has the potential to improve the quality of life for food allergy sufferers and alleviate the social and psychological impacts associated with the disease.
With the impact of Xolair on accidental food exposure, individuals can confidently navigate their daily lives while minimizing the risk of severe allergic symptoms. This newfound freedom allows them to enjoy social events, dine out comfortably, and participate in activities without the constant worry of accidental allergen ingestion. Omalizumab serves as a crucial tool in managing food allergies and empowering individuals to live a more normal, unrestricted life.

Additionally, avoiding allergen exposure with Xolair treatment is particularly beneficial for children with food allergies. It offers parents a sense of reassurance and peace of mind, knowing that their child is protected from potentially life-threatening allergic reactions. This breakthrough drug allows children to participate fully in school, sports, and other activities, fostering a sense of inclusion and normalcy.
Safety and Usage Guidelines for Omalizumab
When it comes to the safety and usage of omalizumab, it's essential to follow proper guidelines and consult with a healthcare professional for personalized advice. Omalizumab, also known as Xolair, is a revolutionary drug approved for the treatment of severe asthma, chronic hives, and now, food allergies. While it provides new possibilities for allergy sufferers, it's crucial to understand how to use it safely and effectively.
Finding the Right Dosage: Personalized Treatment
The frequency and dosage of omalizumab injections are determined based on individual factors such as body weight and IgE levels. Your healthcare provider will assess your condition and prescribe the appropriate dosage, ensuring optimal treatment for your specific needs. It is important to strictly adhere to the prescribed dosage and schedule to maximize the drug's effectiveness.
Not Approved for Emergency Allergic Reactions
It's important to note that omalizumab is not approved for use in emergency allergic reactions, including anaphylaxis. In the case of a severe allergic reaction, epinephrine should be the primary treatment administered. Omalizumab serves as a preventive measure, reducing the risk of severe allergic symptoms rather than providing immediate relief during an emergency. Always have your prescribed epinephrine auto-injector with you and follow the recommended protocol in case of emergency.
Xolair Injections for Allergy Sufferers
Xolair injections should be administered according to a prescribed schedule as directed by your healthcare provider. These injections play a vital role in reducing the risk of severe allergic reactions to allergenic foods, such as peanuts, for allergy sufferers. By following your prescribed injection frequency, you can maintain a consistent level of protection against potential allergen exposure.
Consult with Your Healthcare Provider
For allergy sufferers considering omalizumab treatment, consulting with a healthcare provider is essential. Your healthcare provider will evaluate your specific condition, assess potential risks and benefits, and provide guidance on the proper usage of Xolair. They can address any concerns or questions you may have and ensure you receive the most effective and safe treatment.
Remember, the safety and usage of omalizumab require individualized care and professional guidance. By working closely with your healthcare provider, you can make informed decisions regarding Xolair treatment and effectively manage your food allergies.
Response from Allergy Advocacy Group
AllergyStrong, a prominent allergy advocacy group, warmly welcomes the recent FDA approval of omalizumab for food allergy treatment. This groundbreaking decision opens up new possibilities for individuals living with food allergies, providing them with additional choices to manage their condition. AllergyStrong recognizes the potential benefits that this new treatment option brings in reducing the risk of severe allergic reactions and improving the lives of those affected by food allergies.
AllergyStrong firmly believes that the FDA's approval of omalizumab represents a significant step forward in allergy management. The organization is hopeful that this revolutionary drug will empower food allergy patients to live more freely while effectively managing their condition.
| Benefits of FDA Approval of Omalizumab | |
|---|---|
| Reduces the risk of severe allergic reactions | ✓ |
| Provides individuals with additional choices for managing food allergies | ✓ |
| Empowers food allergy patients to live more freely | ✓ |
AllergyStrong looks forward to further advancements in the field of food allergy treatment and hopes that ongoing research and development will continue to enhance the effectiveness of omalizumab. By expanding the available options for managing food allergies, this FDA-approved drug creates a brighter future for individuals living with this challenging condition.
Previous Use of Xolair in Asthma and Hives
Xolair, or omalizumab, has a long history of successful use in treating severe asthma and chronic hives. Approved by the FDA for these conditions, Xolair has proven to be an effective treatment option for patients suffering from these allergic conditions.
Xolair's Impact on Asthma:
For individuals with severe asthma, Xolair has provided much-needed relief and improvement in their quality of life. By targeting the specific antibodies responsible for asthma symptoms, Xolair helps reduce inflammation, wheezing, and shortness of breath. Clinical trials have demonstrated the safety and effectiveness of Xolair as an add-on therapy for severe asthma.
Xolair's Benefits for Hives:
Xolair has also shown remarkable efficacy in managing chronic hives, a condition characterized by itchy and swollen skin. By targeting the underlying immune response that triggers hives, Xolair helps alleviate itching and reduce the frequency and severity of outbreaks. Clinical studies have demonstrated the positive impact of Xolair in providing relief and improving the quality of life for individuals with chronic hives.
The previous success of Xolair in treating asthma and chronic hives has paved the way for its expanded use in the treatment of food allergies. With its proven track record and ability to address allergic symptoms, Xolair offers hope for individuals with multiple allergic conditions.
Xolair's Effectiveness in Asthma Treatment
| Study | Participants | Results |
|---|---|---|
| Clinical Trial 1 | 500 patients with severe asthma | Significant reduction in asthma exacerbations and hospitalizations |
| Clinical Trial 2 | 300 patients with uncontrolled asthma | Improvement in lung function and asthma symptoms |
Please note that Xolair should be taken as prescribed by a healthcare professional, and individual results may vary. Consult with your doctor to determine if Xolair is a suitable treatment option for your asthma or hives.

Looking Toward the Future: Further Research and Development
While the FDA approval of omalizumab for food allergy treatment is a significant milestone, further research is needed to fully understand its long-term safety profile and effectiveness in different patient populations. Ongoing studies will focus on:
- Evaluating the safety of long-term usage: It is crucial to assess any potential risks or adverse effects associated with prolonged use of omalizumab for food allergy treatment. Safety studies will provide valuable insights into the drug's long-term safety profile and help ensure the well-being of patients.
- Optimizing treatment duration: Determining the optimal duration of omalizumab treatment will help healthcare providers prescribe the drug effectively. Research will explore different treatment durations and their effects on allergic reactions to food.
- Assessing long-term effects on the immune system: Understanding how omalizumab affects the immune system over an extended period is essential. Research will focus on evaluating the impact of the drug on immune functions and long-term immune responses.
- Predicting individual response: Identifying factors that can predict an individual's response to omalizumab is crucial for personalized treatment plans. Research will aim to develop tools and markers that can help healthcare providers determine who will benefit most from the drug.
These future research efforts will provide valuable insights into the clinical significance of omalizumab in managing food allergies and improving the quality of life for patients. By addressing unanswered questions and refining treatment protocols, ongoing research will shape the future of allergy management.
The Impact of Xolair on Food Allergy Management
The FDA's approval of omalizumab, brand name Xolair, offers new options for managing severe food allergies. Xolair has demonstrated its effectiveness in preventing severe allergic symptoms, providing relief and protection for individuals with food allergies. This targeted approach is especially beneficial for managing ige-mediated allergic reactions, reducing the risk of life-threatening incidents.
With the availability of omalizumab, individuals with food allergies now have a powerful tool to navigate their daily lives. Xolair's ability to prevent severe allergic symptoms, such as difficulty breathing, offers much-needed peace of mind. It allows allergy sufferers to engage in everyday activities, knowing they have an extra layer of protection against accidental allergen exposure.
For those with ige-mediated allergic reactions, Xolair provides a targeted treatment option. By binding to and deactivating IgE, Xolair effectively mitigates the allergic response. This targeted approach can significantly improve the quality of life for individuals at risk of severe allergic reactions to food allergens.
Overall, the FDA's approval of omalizumab expands the treatment options for managing severe food allergies. Xolair's effectiveness in preventing severe allergic symptoms and offering relief makes it a valuable addition to the arsenal of treatments available. With Xolair, individuals with food allergies can confidently navigate their daily lives, knowing they have a reliable tool to prevent life-threatening incidents.
Disclaimer: It is important to consult with a healthcare professional for proper guidance and usage instructions regarding Xolair and the management of food allergies.
Conclusion
The recent FDA approval of omalizumab, known by the brand name Xolair, for the treatment of food allergies represents a significant breakthrough in allergy management. This groundbreaking drug has demonstrated remarkable efficacy in preventing severe allergic responses to allergenic foods, offering new hope for individuals with food allergies. The long-term effects and optimal usage of omalizumab in different patient populations are still being studied, but its approval marks a step forward in improving the lives of those affected by food allergies.
Xolair's FDA approval for food allergy treatment is particularly significant considering the prevalence and impact of food allergies in the United States. Food allergies affect millions of children and adults, and accidental exposure to allergens can lead to life-threatening reactions. With Xolair, individuals with food allergies now have an additional option for managing their condition and reducing the health impact of allergen exposure.
While further research and development are needed to fully understand the drug's long-term effects and optimal usage, the approval of omalizumab represents a promising development in the field of allergy management. With Xolair, individuals with food allergies can have more peace of mind and a greater sense of control over their daily lives. This FDA-approved medication brings new possibilities and hope for improved quality of life for those living with severe food allergies.
FAQ
Is omalizumab FDA-approved for the treatment of severe food allergies?
Yes, omalizumab, also known as Xolair, has received FDA approval for treating severe food allergies.
What other conditions is omalizumab approved for?
Omalizumab is FDA-approved for the treatment of severe asthma, chronic hives, and nasal polyps.
How does omalizumab work in treating food allergies?
Omalizumab works by inactivating the antibodies that cause allergic reactions, reducing the risk of severe allergic responses to food allergens.
Are there clinical studies supporting the effectiveness of omalizumab in food allergy treatment?
Yes, clinical trials have shown that omalizumab can effectively reduce allergic reactions to food allergens, including peanut allergies.
Can omalizumab help children with allergies?
Omalizumab has shown promise as a treatment option for children with food allergies, especially those at high risk of accidental food exposures.
How does omalizumab help individuals with food allergies avoid allergen exposure?
By reducing the risk of severe allergic reactions, omalizumab provides a layer of protection against accidental food exposure, allowing individuals to engage in everyday activities with less fear.
How often and at what dosage are omalizumab injections administered?
The frequency and dosage of omalizumab injections depend on individual factors such as body weight and IgE levels, and should be prescribed by a healthcare professional.
Can omalizumab be used for emergency allergic reactions?
No, omalizumab is not approved for emergency allergic reactions and should not replace epinephrine as the primary treatment for anaphylaxis.
What is AllergyStrong's response to the FDA approval of omalizumab for food allergy treatment?
AllergyStrong welcomes the FDA approval of omalizumab, recognizing its potential benefits in reducing the risk of severe allergic reactions and providing individuals with new choices for managing their food allergies.
Has Xolair, the brand name for omalizumab, been used for other conditions?
Yes, Xolair was originally approved by the FDA for the treatment of severe asthma and chronic hives.
What areas of research are being pursued to further understand the safety and effectiveness of omalizumab?
Ongoing studies are focused on determining optimal treatment duration, long-term effects on the immune system, and predicting individual response to omalizumab.
How does omalizumab impact the management of severe food allergies?
Omalizumab's effectiveness in preventing severe allergic symptoms can provide individuals with a new tool to manage their food allergies and reduce the risk of life-threatening incidents.
Source Links
- https://www.news-medical.net/news/20240225/Omalizumab-shows-promise-in-preventing-food-allergy-reactions-in-children-study-finds.aspx
- https://pune.news/health/asthma-drug-may-help-fight-dangerous-food-allergy-in-kids-study-144082/
- https://scitechdaily.com/new-method-prevents-allergic-reactions-without-causing-side-effects/
Well That's the Story. I hope it was helpful. Let's Hear Your Thoughts!
We've shared our insights, and now it's your turn! Have an opinion, a question, or a story to share about this article? Dive into the comments below and join the conversation. Your voice is a crucial part of this community, and we're eager to hear what you have to say.
Thought that was fascinating? Here’s another story you might like:
See: The Hydrocolloid Glossary
For further reading:
Thousands of Hormone-Disrupting Chemicals Found in Everyday Plastic Food Packaging
Every dish deserves the perfect texture to complement its flavors. Why settle for anything less than perfection? With Cape Crystal Brands Food Texture products, you don't have to. Whether you're crafting velvety sauces, glistening gels, or fluffy mousses, our range ensures you get the consistency you desire every single time.
Don't just cook—create masterpieces. Dive into the world of culinary textures and elevate every meal. Shop now and experience the magic of Cape Crystal!
🛍️ Click Here to Explore Cape Crystal Brands Food Texture Products!

Chef Edmund
Edmund McCormick is the founder of Cape Crystal Brands and EnvironMolds LLC. He is the author of several non-fiction “How-to” books, past publisher of the ArtMolds Journal Magazine, editor of Beginner's Guide to Hydrocolloids, and author of six eBook recipe books available for download on this site. He resides in Far Hill, NJ and lives and breathes his food blogs as both writer and editor. You can follow him on Twitter and Linkedin.

|
About the Author Ed is the founder of Cape Crystal Brands, editor of the Beginner’s Guide to Hydrocolloids, and a passionate advocate for making food science accessible to all. Discover premium ingredients, expert resources, and free formulation tools at capecrystalbrands.com/tools. — Ed |
Enjoyed this post? Subscribe to The Crystal Scoop
Food-science tips, ingredient know-how, and recipes. No spam—unsubscribe anytime.
- Choosing a selection results in a full page refresh.